New device reduces stroke risk in A-fib patients
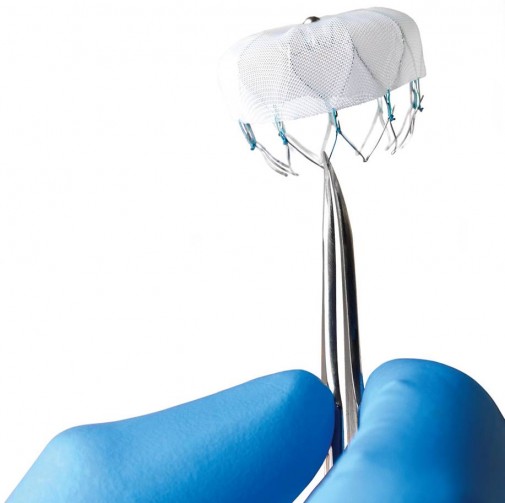
A small device that prevents stroke in people with atrial fibrillation (A-fib) is now available for implantation after receiving approval by the U.S. Food and Drug Administration in March 2015.
The WATCHMAN device, which is inserted through a minimally invasive catheterization procedure, seals off the left atrial appendage (LAA) where blood clots can develop and travel from the heart to the brain, triggering a stroke.
“Studies show that patients with A-fib are five times more likely to suffer a stroke than someone without the heart condition,” says Dr. Erik Wissner, a cardiologist at the Advocate Heart Institute on staff at Christ Medical Center in Oak Lawn, Ill. “With more than 90 percent of blood clots forming in the left atrial appendage, the risk of stroke is significantly reduced when the WATCHMAN device permanently closes off that area.”
A-fib patients have long been treated with anticoagulants, blood-thinning medications, which reduce the risk of stroke but pose many serious risks like excessive bleeding.
“Those who are especially at risk for excessive bleeding while taking anticoagulants include elderly patients who are at a high risk for falls or people who lead very active lifestyles,” explains Dr. Wissner. “While taking blood-thinning medications, patients often have to change their lifestyle or constantly worry about internal bleeding from a seemingly minor fall or bump.”
The WATCHMAN device gives people a piece of mind and allows them to continue their normal activities while reducing their risk of stroke, he says.
Dr. Wissner, who has performed the procedure for the past six years in Germany, where the device had previously been approved, says the implantation is minimally invasive and completed in approximately 45 minutes. While the patient is under general anesthesia, cardiologists use a catheter to direct the quarter-sized device, which resembles a small mesh parachute, through a leg vein and up to the heart for it to be implanted.
Patients typically go home after an overnight stay and are able to return to normal activities within two days and sports after 10 days.
“Many people will benefit from this breakthrough device,” says Dr. Wissner. “This is a safe and effective alternative to taking anticoagulants.”
Advocate Christ Medical Center is now a WATCHMAN implantation center.
Related Posts
Comments
About the Author
Julie Nakis, health enews contributor, is manager of public affairs at Advocate Children's Hospital. She earned her BA in communications from the University of Iowa – Go Hawkeyes! In her free time, she enjoys spending time with friends and family, exploring the city and cheering on the Chicago Cubs and Blackhawks.















